Rita H. Khoury, MD; BP. Salmon, MS; Asha Gandhi, BS; Prital K. Patel, BA; Peter Gudaitis, BA; Dauna Gudaitis, BA
(2011) Abstracts and Case Studies From the College of American Pathologists 2011 Annual Meeting (CAP ’11). Archives of Pathology & Laboratory Medicine: September 2011, Vol. 135, No. 9, pp. 1102-1230.
Context: Clostridium difficile (C.Diff) is a gram-positive anaerobe producing enterotoxin and cytotoxin (toxin A and B, respectively), toxin B, seemingly, essential for the virulence of C.Diff. It is responsible for 15% to 25% of antibiotic-associated diarrhea and 95% to 100% of antibiotic-associated pseudomembranous colitis, and it is one of the most widespread infections acquired in long-term care facilities. The tests available for C.Diff confirmation include enzyme immunoassays, tissue culture cytotoxicity, anaerobic culture, glutamate dehydrogenase antigen, and nucleic acid amplification.
Design: Eighty-nine stool specimens were collected from residents in long-term care facilities with symptoms of C.Diff colitis. All samples were tested simultaneously by the same technician with Premier Toxins A and B (enzyme immunoassay) and the GeneOhm assay, real-time polymerase chain reaction that detects toxin B (tcdB) gene. Discordant samples were sent for confirmation by toxigenic culture.
Results: Of the patients, 57.3% were female, 26 patients were positive, and 63 negative in the Premier assay, whereas 32 were positive and 56 negative in the GeneOhm assay (Table). Ten discordant samples were sent for toxigenic culture and the results confirmed GeneOhm assay results.
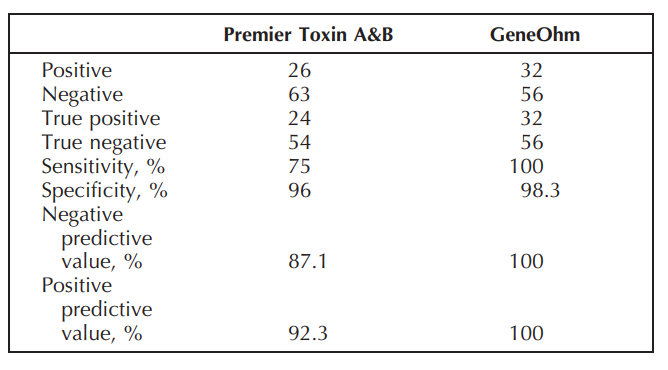
Conclusion: The GeneOhm assay offers better sensitivity and specificity over the enzyme assay to diagnose C.Diff. In addition, it allows for early treatment and appropriate infection control. Although the initial cost is higher, if we take into consideration the cost of unnecessary repeat testing due to a lack of physician confidence in the results, and the cost of unnecessary isolation due to false positive results, the final cost will be much lower for the facilities/hospitals than enzyme immunoassay.


Leave A Comment
You must be logged in to post a comment.