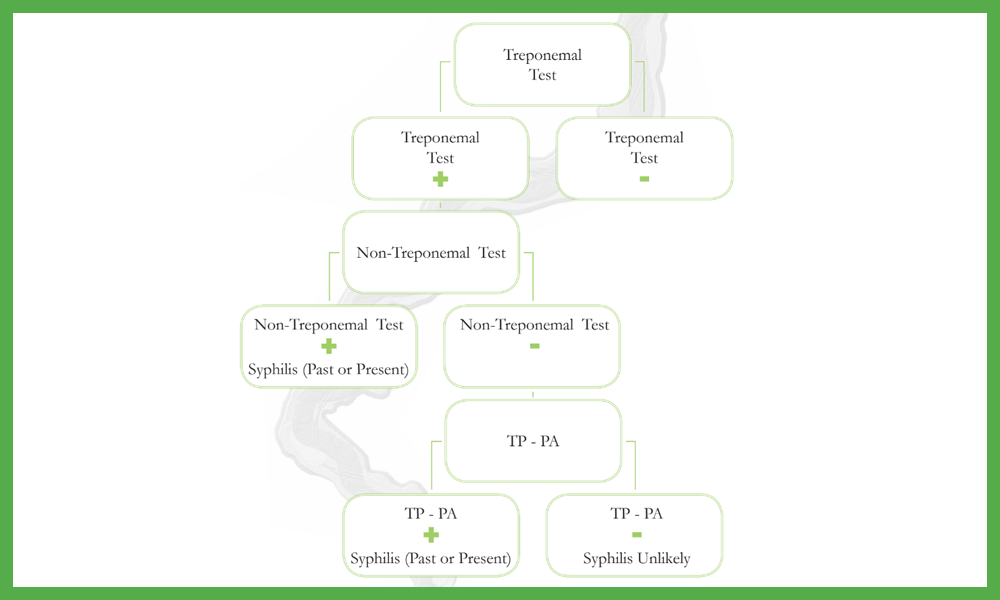
Aculabs Lab Director Presents Paper on Vitamin D Ordering at CAP25
Aculabs is proud to share that our laboratory director, Rita Khoury, MD., recently had the opportunity to present several papers at the College of American Pathologists (CAP) 2025 meeting. While at the conference, Dr. Khoury sat down and spoke about her findings regarding Vitamin D testing with Medscape, a leading healthcare publication. "Orders for